
“The press discharges vital functions as a bloodhound as well as a watchdog.”
— Lord Nicholls in Reynolds v Times Newspapers, October 1999
Brian Deer’s Sunday Times investigation
[Long-read summary: 4,000 words]
With revelations scattered across nearly two decades, Brian Deer, pictured [here], pursued a landmark public interest investigation for The Sunday Times of London — with spin-offs for a TV network, a leading medical journal, and an award-winning book — into the origin of allegations linking a mainstay of medicine to brain and gut damage in children.
These claims concerned a vaccine against measles, mumps, and rubella (MMR). And at the time Deer began, with an empty reporters’ notebook, Britain had been gripped by years of controversy over the three-in-one shot’s safety — spawning fear, guilt, and infectious disease, exported to the United States and many other countries to fuel every kind of confusion over vaccines.
British newspaper headlines of those days powered the scare — too many to quote more than examples:
NEW TESTS ‘CONFIRM THE LINK’ BETWEEN MMR AND AUTISM
— Sunday Express
THE QUESTION EVERY PARENT IS ASKING
— Daily Mail
CRYING SHAME OF THE VACCINATION VICTIMS
— The Sunday Times
Incredibly, the prime source for such concerns — which in the 2020s would go on to seed global “anti-vax” networks opposing covid-19 shots — was a mere five-page research paper published in February 1998 that sought to blame MMR for autism.
Written by a then-forty-one-year-old academic research surgeon, Andrew Wakefield, co-authored by a dozen associates (who played no part in writing it) and published in the prestigious Lancet medical journal, this paper reported on the anonymised cases of just twelve children with apparent developmental issues — most of whose parents were said to finger MMR.

Backed by a twenty-minute video news release and a breathless press conference organised by Wakefield’s employers at the Royal Free Hospital Medical School in Hampstead, North London, the four-thousand-word paper provoked heavy media interest and was followed by a campaign he led.
This included further Wakefield publications attacking MMR, leading to a collapse in public confidence in the shot, which, since the late 1980s in the UK (and the early 1970s in the US) has been given routinely to children at, or soon after, one year of age — eliminating measles and rubella from developed countries.
READ HOW DEER’S WAKEFIELD
INVESTIGATION WAS CHECKED
The loudest alarm centred on claims in the 1998 paper that the parents of two thirds of the twelve kids blamed MMR for what Wakefield called a “new syndrome.” This, he said, combined what he claimed to be a previously unrecognised inflammatory bowel disease and what he called “regressive autism”: a severe disability in which language and basic skills may be lost.
Most disturbingly, Wakefield’s paper described what he called “behavioural” or “neuropsychiatric” symptoms as appearing suddenly after MMR: an average of just 6.3 days following vaccination, with a maximum (in two cases) of 14 days.
As he explained in a patent application discussed later in this summary:
“Before vaccination, the infants were shown to have a normal developmental pattern but often within days of receiving the vaccination some infants can begin to noticeably regress over time leading to a clinical diagnosis of autism.”
Although The Lancet report involved only a dozen White kids, aged between three and nine, and neither its methods nor its results would ever be replicated, medical breakthroughs often begin with small-scale observations. And, if true, such findings might have been the first snapshot of an unrecognised epidemic of devastating injuries to children.
Wakefield didn’t hesitate to promote that suspicion. “It’s a moral issue for me,” he announced at the press conference, where he called for the government to suspend MMR in favour of breaking the triple vaccine into single measles, mumps, and rubella shots to be spaced at yearly intervals. “I can’t support the continued use of these three vaccines, given in combination, until this issue has been resolved.”
Nothing in the paper evidenced this proposition. And the journal’s editors inserted a warning: the research didn’t “prove” any connection. But as the doctor campaigned — with mass media backing — UK vaccination rates spiralled down below a level needed to keep measles at bay.
Even Tony Blair, then the British prime minister, became embroiled in the affair when Wakefield supporters insinuated — the Blairs say wrongly — that one of the politician’s sons never received MMR.
Meanwhile in America, a ferocious anti-vaccine movement took off after Wakefield toured autism conferences and in November 2000 appeared on the CBS network’s 60 Minutes programme linking MMR with what he called an “epidemic of autism”. This was followed by campaigners’ allegations that all vaccines are suspect: either due to their ingredients or because of the growing number recommended.
“In 1983 the shot schedule was ten. That’s when autism was one in 10,000. Now there’s 36, and autism is one in 150,” argued American actor Jenny McCarthy, who at the time blamed MMR for her own son’s autism and for a while gained the top US profile for such beliefs. “All arrows point to one direction.”
Andrew Wakefield unmasked
Journalists (including The Sunday Times‘s) scrambled to showcase Wakefield’s warnings and to spotlight parents’ fears. But in September 2003, reporter Brian Deer was assigned to investigate the controversy and, although not initially focused on Wakefield at all [see this url’s original and unchanged “mmr/lancet-summary” filename], he unearthed a scandal of astounding proportions. Far from the scare being based on honest research, Deer uncovered a monstrous deception.
Unknown even to close Wakefield associates, such as the paper’s senior author John Walker-Smith, the doctor had been secretly payrolled to create evidence against the shot and — while planning business schemes to personally profit from the controversy — had changed and misreported the children’s histories and diagnoses to rig his results in The Lancet.

Before Deer’s intervention, Wakefield had appeared to all the world to be a respectable independent researcher. Tall and square-headed, with hooded eyes and a booming voice, he was the son of doctors (a neurologist and a family practitioner), had grown up in Bath, a prosperous west-of-England spa town, and joined the Royal Free in November 1988 after postgraduate training in Toronto, Canada.
His demeanour was languid — he was privately educated — and, born in September 1956, he seemed nothing less than a lingering example of the presumed honour of the upper middle class.
But Deer’s investigation — nominated in February 2011 for two British Press Awards — discovered that, while Wakefield held himself out to be a dispassionate scientist, two years before The Lancet paper was published — and before any of the children were even seen at the hospital — he’d been hired to attack MMR by a lawyer named Richard Barr: a mediocre jobbing solicitor in the small eastern English town of King’s Lynn, Norfolk, who was attempting to raise a speculative class action lawsuit against the drug companies that made MMR.
Unlike expert witnesses, who give professional advice and opinions, Wakefield had secretly negotiated an unprecedented contract with Barr, then aged forty-eight, to conduct “clinical and scientific research”. Their goal was to find evidence of Wakefield’s claimed “syndrome” which they’d contrived as the centrepiece of (later failed) litigation on behalf of some 1,600 British families — mostly recruited through stories planted in media.
This publicly undisclosed role for Wakefield created the grossest conflict of interest. And the exposure of it by Deer, in February 2004, led to public uproar in the United Kingdom that would resonate around the world for decades. As the Independent newspaper commented in an editorial on the day of Deer’s first Sunday Times report:
“It may turn out that the greatest recent crisis in public health need never have happened.”
Wakefield denied anything was wrong. But within weeks of the first disclosures, almost all his co-authors were so shocked by what they learnt from the Sunday Times reports, they retracted The Lancet paper’s “Interpretation”, or conclusions, section in which MMR and autism were linked. And (after asking to see the journalist’s evidence) from July 2007 to May 2010 the UK doctors’ regulator, the General Medical Council (GMC), held its longest-ever misconduct hearing, reinvestigating Deer’s early findings.
The Barr-Wakefield deal
Despite Wakefield’s talk of his “moral issue,” he was Richard Barr’s hired gun for eight years. Under an undisclosed agreement between the pair, the doctor was paid to make a case against the vaccine with money the lawyer obtained from the UK legal aid fund — run by the government to give less well-off access to justice.
Wakefield charged through Barr at the rate of £150 an hour (about £360 or US$480 an hour at October 2025 values) — billed via a company of Wakefield’s then-wife, Carmel O’Donovan. Payments to him eventually totalled what the UK Legal Services Commission, pressed by Deer under a new freedom of information act, said was £435,643 (more than £800,000, or nearly $1 million, at 2025 values) plus expenses.
These hidden fees — revealed in The Sunday Times in December 2006 — gave the doctor a direct personal, but undeclared, financial interest in his research claims: totalling more than eight times his reported annual salary and creating an incentive for him not only to launch the alarm over the vaccine’s safety but to keep it going for as long as he could.
Even close Wakefield colleagues didn’t know of his deal with Barr, as one of several co-authors of the Lancet study admitted in February 2004. He told Sunday Times health editor Lois Rogers:
“I am very, very angry. I would never have put my name to the study if I had known there was this conflict of interest, and had I not done so it would never have got published.”
In addition to the personal payments, Wakefield had been awarded an initial £55,000 (about £132,000 at 2025 values) for which he’d applied to the Legal Aid Board with Barr in June 1996 (six weeks before the first of the twelve children was admitted to the Royal Free) but which, like the hourly fees, he never declared to the Lancet to carry out the research submitted to the journal.
This start-up funding was part of a staggering £26.2 million of British taxpayers’ money (about £49.3 million, US $65.8m, at 2025 values) eventually shared among a small group of doctors and lawyers working under Barr’s and Wakefield’s direction trying to prove MMR caused his so-called “syndrome”.
Yet more surprising, Wakefield asserted the existence of such a syndrome — allegedly including what he would later dub “autistic enterocolitis” — before he performed the research with which he supposedly discovered it.
This Barr-Wakefield deal was the foundation of the vaccine crisis (morphing into campaigns against shots for everything from human papillomavirus to SARS-CoV-2) both in Britain and throughout the world.
By 2025, the paper’s impact even reached the White House — as Donald Trump made clear in September of that year. “The MMR, I think should be taken separately,” he announced at a televised press conference with Wakefield acolyte Robert Kennedy Jr. “This is based on what I feel. The mumps, measles and, and, er, the three should be taken separately. And it seems that when you mix them there could be a problem. So there’s no downside in taking them separately. In fact, they think it’s better.”
But Trump wouldn’t have grasped where it all began, or known that in August 1997 — six months before The Lancet published Wakefield’s paper — the small-time lawyer Barr had instructed Wakefield:
“The prime objective is to produce unassailable evidence in court so as to convince a court that these vaccines are dangerous.”

And if a mission like that wasn’t enough to cast doubt on Wakefield’s objectivity, Deer unearthed another shocking conflict of interest. In June 1997 — nearly nine months before the press conference at which the then-doctor called for single vaccines — he’d covertly filed for a patent on products, including his own supposedly “safer” single measles vaccine, which could only have stood the slightest prospect of success if public confidence in MMR was damaged.

Although Wakefield later falsely denied the existence of such patent claims, his proposed shot and a network of companies through which he intended to raise money from naïve investors (for purported Wakefield inventions including “a replacement for attenuated viral vaccines”, commercial testing kits, and what he claimed to be a possible “complete cure” for autism) were set out in confidential documents.
One Wakefield business was awarded £800,000 (about £1.49 million, US $1.9 million at 2025 values) from the UK legal aid fund on the strength of (later discredited) data which he’d co-authored with an Irish pathologist named John O’Leary. And, even as The Lancet paper was being prepared, behind the scenes Wakefield was negotiating plans to exploit the public alarm with secret schemes to line his pockets.
“Disgraced doctor Andrew Wakefield plotted to make £28 million a year [£58.2 million or US $77.7 million at 2025 values] from the MMR jab panic he triggered,” was how the British newspaper The Sun, for example, reported in January 2011 on this disclosure from Deer.
Behind the veil
As with the researcher, so too with his subjects. They also weren’t as they appeared. The twelve children (eleven boys and one girl) reported in The Lancet were held out as if merely a routine series of developmentally challenged patients with bowel symptoms needing investigation by the Royal Free Hospital’s gastroenterologists.
That so many of the kids’ parents were said to blame autism on the same common vaccine, understandably, caused widespread concern.
But Deer discovered that nearly all the children had been pre-selected through MMR campaign groups, and that, at the time of their admission to the hospital, most of their parents weren’t only clients or contacts of Richard Barr’s law firm but had been coached by Wakefield in phone calls to their homes before they saw the Royal Free’s clinicians.
None of the twelve lived in London. Two were brothers. Two attended the same doctors’ office, hundreds of miles from the Royal Free. Three were patients at another clinic. One was flown in from the United States.
Deer’s investigation revealed, moreover, that the paper’s pivotal purported finding — the onset of autism symptoms within days of vaccination — was a sham: laundering into medical literature, as apparent facts, the unverified — and sometimes altered — assertions of a group of unnamed parents who (unknown to the journal and its readers) were bound to blame MMR when they came to the hospital because that was why they were invited.
Доктор, который одурачил весь мир

Parents blaming the shot wasn’t a “finding” at all. It was a qualification to take part in the research.
For years, Wakefield — a former trainee gut surgeon, contractually forbidden at the Royal Free from providing care to patients — denied this. But confidential medical records revealed the true number of children whose parents claimed, suspected, or feared were injured by MMR wasn’t eight, as the paper reported: it was eleven of the twelve — later all twelve — with key case records noting legal involvement before kids were referred to the hospital.
Regarding the only little girl in the project, for instance (who didn’t, as Wakefield evidently suggested with the mention of Crohn’s) have inflammatory bowel disease, her local family physician noted:
“Mum taking her to Dr Wakefield, Royal Free hospital, for CT scans, gut biopsies, ?Crohn’s. Will need referral letter. Dr Wakefield to phone me. Funded through legal aid.”
In the light of such discoveries, the grounds were overwhelming to dig deeper into Wakefield’s activities. In an exercise never before accomplished by a journalist, Deer was able to gain parents’ co-operation, attend the GMC hearing (reinvestigating his first findings) and go behind the face of the 1998 paper to prove what really went on.
Penetrating veils of medical and legal confidentiality, Deer discovered the hospital’s clinicians and pathology service found nothing to implicate MMR, but Wakefield (who alone wrote The Lancet paper) had falsified diagnoses and patient histories to make it appear he’d found his “syndrome” when he hadn’t.
As first revealed by Deer in The Sunday Times in February 2009, the effect was to give the impression of a link between MMR, bowel disease, and the sudden onset of autism when otherwise none was evident.
Not only did Wakefield alter what he called kids’ “behavioural” or “neuropsychiatric” diagnoses but he suppressed blood tests for bowel disease after they came back normal and misrepresented commonplace observations in children’s guts (including “ileal lymphoid nodular hyperplasia”, part of The Lancet paper’s title) as purported evidence of the syndrome he sought.
What the hospital’s clinicians and pathology service actually found in the patients — but Wakefield didn’t report in The Lancet — was severe constipation with overwhelmingly normal gastrointestinal biopsies and benign or normal features.
When taken together with misrepresented developmental histories, not one case was free of critical mismatches between the twelve-child paper and the patients’ contemporaneous records. Some kids were a cause for concern before vaccination. Some were deemed normal months afterward. Some weren’t diagnosed with autism at all.
Among the children falsely reported as suffering from Wakefield’s made-up “syndrome” (which even decades later wouldn’t be recognised in medical literature) was the penultimate case in the series: a five-year-old boy from northern California whose father was outraged by the deception.
After studying the paper, that father wrote to Deer:
“If my son really is Patient 11, then The Lancet article is simply an outright fabrication.”
Protections sidelined
In addition to discovering the study was rigged, Deer uncovered a raft of further issues including false claims of ethical supervision.
Research on patients is governed by national and international standards — particularly the Helsinki Declaration [1996 revision] — and no reputable hospital review board would have endorsed the kind of fishing expedition into the guts of young children that Wakefield embarked on for Barr. Without that endorsement, moreover, no reputable journal would have published any resulting paper.
布莱恩-迪尔 | 欺骗世界的
Against that background, to satisfy The Lancet‘s patient-protection requirements, but without revealing to hospital authorities what was really going on, Wakefield dishonestly reported that a gruelling five-day battery of invasive and distressing procedures he caused to be performed on the kids — including anaesthesia, ileocolonoscopies, lumbar punctures, MRI brain scans, EEGs, radioactive drinks and x-rays — pre-agreed with Barr for the lawsuit, was approved by the Royal Free’s ethics committee.
As The Lancet paper stated under the heading “Ethical approval and consent”:
Investigations were approved by the Ethical Practices Committee of the Royal Free Hospital NHS Trust, and parents gave informed consent.
But Deer proved that, despite the research being executed on the uniquely vulnerable, developmentally challenged children of sometimes distraught parents hoping for money to help with their care, the ethics committee wasn’t told the truth about the project and never gave any such approval.
Responding to Deer in February 2004, Wakefield and his key accomplices — Royal Free paediatricians and Lancet co-authors John Walker-Smith and Simon Murch — denied this explosive discovery and issued a formal statement for media. But, after being confronted with the evidence at the GMC hearing, they changed their story and — despite clear rules — now argued they needed no approval.
And yet in The Lancet they’d expressly claimed to have it: requiring a London High Court judge to intervene. As Justice Mitting later ruled of the ethics statement (written by Wakefield):
“This statement was untrue and should not have been included in the paper.”
Andrew Wakefield’s science
Deer also probed Wakefield’s basic science. The story was much the same. The doctor obtained the legal money and planned his business ventures against a fringe hypothesis of his own that the culprit for both the (fake) inflammatory bowel disease and (unsubstantiated) regressive autism was persistent infection with measles virus — an essential component of MMR.
But Deer demonstrated on UK TV that sophisticated, unreported, molecular tests validated and carried out in Wakefield’s own lab, had found no trace of measles in the children’s guts, blood, or cerebrospinal fluid. Those tests, moreover, were among a string of findings from laboratories around the world which found no association between the virus and autism.
In a further report by Deer, The Sunday Times also disclosed critical flaws in one apparently positive study produced in Dublin by John O’Leary. Using biological samples supplied by Wakefield, this misled thousands of families in the UK and US, ensnared for years in hopeless litigation almost entirely based on Wakefield’s claims.
Deer (who in April 2006 reported the first British measles death in 14 years) took no view on whether vaccines may or may not cause autism. But, tellingly, Wakefield refused his institution’s repeated written requests to replicate his research with more child patients and with blinded scientific testing — a refusal triggering his dismissal from the Royal Free.
In the decades that followed, Deer never found any scientific material repeating The Lancet project or its findings. Although, self-evidently, all kinds of children, including those with autism, suffer from constipation and other digestive issues, he learnt of a mass of authoritative research comprehensively contradicting Wakefield.
“Specifically, numerous studies have refuted Andrew Wakefield’s theory that MMR vaccine is linked to bowel disorders and autism,” was how the American Academy of Pediatrics summarised the consensus in an August 2009 statement to NBC News for a Dateline programme on the clash between Wakefield and Deer. “Every aspect of Dr Wakefield’s theory has been disproven.”
The English courts would also speak on this aspect — for example in the ruling of Justice Mitting, handed down in March 2012:
“There is now no respectable body of opinion which supports his hypothesis, that MMR vaccine and autism/enterocolitis are causally linked.”
And England’s Court of Appeal declared in May 2020:
“Dr Wakefield’s paper has since been wholly discredited.”
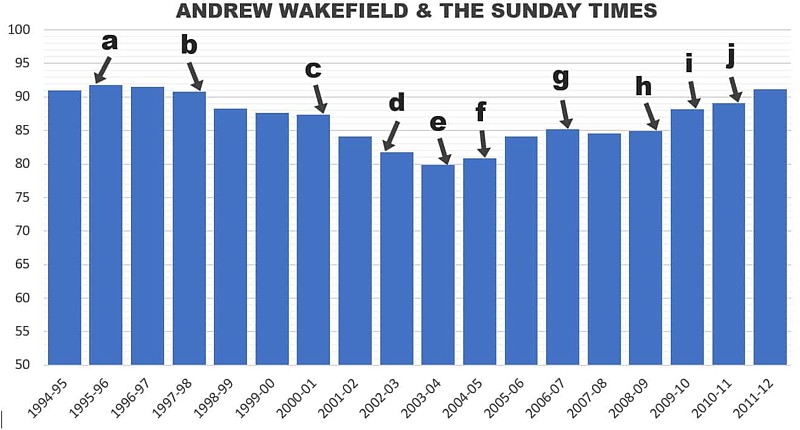
The impact of Deer’s investigation has been felt around the world, with media coverage from New Zealand to Canada. In the UK, The Sunday Times revelations prompted huge follow-up by other news outlets, and (after years during which MMR vaccination rates fell in response to Wakefield and Barr’s campaign) a reversal in the trend over the years of Deer’s reporting until public confidence was fully restored.
READ HOW DEER’S WAKEFIELD
INVESTIGATION WAS CHECKED
Meanwhile, in the US — where the Barr-Wakefield deal was joined by unsubstantiated complaints marshalled by then-product liability lawyer Robert Kennedy Jr and personal injury attorneys that a mercury-based vaccine preservative, thimerosal, was also a cause of autism — findings by Deer were presented by the Department of Justice in federal court, followed in February 2009 by scathing rulings.
After hearing a test case of petitions from some 5,000 US families, one presiding special master (a specialist judge) said:
“It is a noteworthy point that not only has that ‘autistic enterocolitis’ theory not been accepted into gastroenterology textbooks, but that theory, and Dr Wakefield’s role in its development, have been strongly criticized as constituting defective or fraudulent science.”
Wakefield denies everything
In response to Deer, Wakefield supporters falsely denied he took money for research or claimed his own vaccine. And, amid a barrage of sometimes paid-for smears, a two-decade campaign of malicious libel by an ex-lawyer named Clifford Miller, and crank abuse of the journalist by such individuals as one Carol Stott and Martin Walker (all working with Wakefield) who lauded the doctor as a “hero”.
The father-of-four Wakefield’s deceits, however, hadn’t only triggered the resurgence of sometimes fatal or brain-disabling diseases. It plunged countless parents into the hell of believing it was their own fault for agreeing to vaccination that a son or daughter developed autism. He’d also misled an ethics committee over child rights and safety, betrayed a vaccine safety whistleblower whose identity he discovered, and bought blood from children as young as four years old, attending a birthday party, then joked about them crying, fainting and vomiting.
Get Deer’s book: The Doctor Who Fooled the World
Meanwhile, Wakefield denied any conflicts of interest. Instead of admitting the obvious, he initiated a retaliatory smear campaign and even claimed never to have said that MMR caused autism in the first place. But documents — including patents — conclusively evidence his assertions at the time. And he published a string of further falsified reports to undermine confidence in the shot.
Even when he knew his allegations were proven baseless, he was found promoting them from a controversial business in Austin, Texas, called Thoughtful House, where — after being fired from the Royal Free in October 2001 following his express refusal to validate The Lancet study with rigorous science — he held a lucrative post spun from his campaign.

From the outset of Deer’s investigation, Wakefield refused to be interviewed, filed baseless complaints, and issued statements denying every aspect. He also launched, sought to stall, then abandoned with some £1.3 million (about $1.7m) costs (more than £2.2 million, US $2.9m, at 2025 values), a two-year “gagging” libel lawsuit in London, financed by the Medical Protection Society which defends doctors against their patients.
In reply, Deer pressed the court for a speedy trial to clear his name, publicly accusing Wakefield of being “unremittingly evasive and dishonest”.
The doctor’s conduct in this litigation was also damned by the judge hearing the case, who said Wakefield appeared to have filed suit for “public relations purposes,” “wished to extract whatever advantage he could from the existence of the proceedings while not wishing to progress them,” and was using them as “a weapon in his attempts to close down discussion and debate over an important public issue.”
Wakefield paper retracted
Faced with overwhelming proof of grotesque misconduct, Wakefield denied rigging his results and concocted a preposterous conspiracy theory (the first of several) to account for his exposure. In a March 2009 statement, he went on to argue:
“The notion that any researcher can cook such data in any fashion that can be slipped past the medical community for his personal benefit is patent nonsense. Scientific rigor requires repeatability for verification of any research and Mr Deer’s implications of fraud against me are claims that a trained physician and researcher of good standing had suddenly decided he was going to fake data for his own enrichment.”
Yet that’s precisely what Wakefield did. And on 28 January 2010 — after 217 days of evidence and submissions — a panel of three doctors and two lay members (a statutory tribunal under the UK Medical Act 1983) hearing the GMC case handed down verdicts condemning Wakefield’s “serious professional misconduct” and one hundred percent vindicating Deer.
Branding Wakefield “dishonest”, “unethical” and “callous”, the panel found him guilty (against the criminal standard of certainty) of some three dozen charges, including four counts of dishonesty (including one Wakefield himself submitted through counsel to be a charge of “fraud”) and twelve involving the abuse of developmentally challenged children.
The panel found his research to be dishonest and performed without ethical approval. And, five days after these rulings, The Lancet fully retracted the 1998 paper from the scientific literature as “utterly false”, prompting international media interest and further retractions.
“What is indisputable is that vaccines protect children from dangerous diseases,” said The New York Times, in one of a string of editorials in leading newspapers. “We hope that The Lancet’s belated retraction will finally lay this damaging myth about autism and vaccines to rest.”
Three weeks later, on 17 February 2010, Wakefield was ousted by the directors of his Thoughtful House business in Texas, and on 24 May — day 217 of the GMC hearing — he was ordered to be erased from the register of UK doctors, ending his career in medicine.
Journal calls the fraud
Although, like his co-defendants in the GMC proceedings, Wakefield was insured by the Medical Protection Society, on 21 December 2010 (after his legal team withdrew support for his case) his erasure from the register was confirmed and made permanent when he abandoned an appeal against the verdicts.
But this extraordinary story of deceit and betrayal would continue even after he was finished in medicine. So, in January 2011, the British Medical Journal (BMJ), commissioned Deer to reprise his investigation for a medically qualified readership with a three-week package of disclosures.
That short series of lengthy reports (including peer review, editorial checking, and sixty hours of legal review) was endorsed in an editorial calling Wakefield’s twelve-child study “fraudulent” and accusing the Royal Free medical school and The Lancet of institutional and editorial “misconduct”.
Regarding Deer, the BMJ‘s editors noted a paradox. A mere reporter had beaten experts to the truth:
“It has taken the diligent scepticism of one man, standing outside medicine and science, to show that the paper was in fact an elaborate fraud.”
Among hundreds of reports worldwide on Deer’s revelations in the BMJ — which were covered by all north American networks and reached almost half of Americans surveyed days later in a Harris poll — The New York Times said in a second editorial on the affair:
“Now the British Medical Journal has taken the extraordinary step of publishing a lengthy report by Brian Deer, the British investigative journalist who first brought the paper’s flaws to light — and has put its own reputation on the line by endorsing his findings.”
Three months later, Deer’s personal journey reached a turning point when in April 2011 he was nominated as Britain’s Reporter of the Year, and named Specialist Journalist of the Year, in the British newspaper industry’s annual Pulitzer-style Press Awards. Judges for the Society of Editors praised what they called his “outstanding perseverance, stamina and revelation on a story of major importance,” and said of his investigation:
“It was a tremendous righting of a wrong”.
Parents speak out
And yet still Wakefield refused to show regret and, in a bid to raise money by tricking his supporters, the following year filed another vexatious lawsuit: this time in Austin, Texas. Like his London action, however, it stood no prospect of success and was thrown out with costs awarded against him.
His then-wife, Carmel O’Donovan, admitted later: “The minute the judge walked in I thought we are scuppered.”
In fact, it appeared the Texas case was kept alive to create misleading content for a faux “documentary.” This was the work of one Miranda Bailey who, in a contrived collaboration with Wakefield, faked the appearance of “fly on the wall” coverage at his Austin home and lied that Deer refused to reply.
READ HOW DEER’S WAKEFIELD
INVESTIGATION WAS CHECKED
More credible sources made the position clear, such as the United States National Institutes of Health. In an August 2017 response to the anti-vaccine campaigner Robert Kennedy Jr, the institutes’ director and senior managers wrote:
“The claimed causal connection between childhood vaccines and autism, based on an initial British study that has conclusively been shown to be fabricated, has been exhaustively studied in hundreds of thousands of children and resoundingly rejected.”
That might have concluded many years of inquiry: a rarely paralleled newspaper investigation. As Deer summarised from a position of the utmost confidence:
This story is so detailed and my reporting so persistent, moreover, that either it’s honest and correct — in substance and sting — or Wakefield must be victim to the most sustained barrage of malicious defamation in the history of the English language.
But if the latter were the case, this acknowledgement could expose me to disgrace and bankruptcy, with my publishers contractually entitled to pulp my book and make me pick up the bill.
Meanwhile, the ex-doctor’s dishonesty provoked further developments as parents, former colleagues, and others came forward to tell the journalist what they knew.
One mother, who for years betrayed Wakefield’s network from the inside, declared of her reasoning for secretly collaborating with Deer:
“I had a situation where there were two men… One of them was saying ‘white’ and the other ‘black’. One of them was an honourable man. And one of them was a shyster.”
Finally, in September 2020 (delayed by the pandemic) came Deer’s exhaustively and independently checked book, The Doctor Who Fooled the World. This was published in North America by Johns Hopkins University, elsewhere in English by the quality publishers Scribe, and later in Chinese, Russian, and other languages.
Among reviews, the top science journal Nature described the book as “riveting,” The Times called it “remarkable,” and The Wall Street Journal explained:
“Exposing researchers who lie, cheat and fake their data often requires the work of courageous whistleblowers or tenacious investigative journalists. Enter Brian Deer.”
Among many new revelations regarding what the book calls “a conspiracy of fraud and betrayal,” it identifies not only the sham science behind the attack on MMR, with critical information withheld from the public for decades, but also the horrifying suffering of children in Wakefield’s research, some held down kicking and screaming as they resisted the battery of unethical experiments he caused to be forced upon them.
And then the upshot: exquisite final evidence in the words of another of The Lancet twelve’s parents. Coming forward to supply Deer with a trove of contemporaneous documents from the 1990s — proving Wakefield’s misconduct all over again — she wrote:
“I know that paper is not right and fraudulent. I can see that from what was written about my son.”
READ HOW DEER’S WAKEFIELD
INVESTIGATION WAS CHECKED



Invite Brian Deer to speak at your event
Hear the astonishing true story of the plot behind today’s controversy over vaccines, and its exposure by an investigative journalist
